Japan Early Access Partner | Supplying Drugs Before Approval
Contact Us
Free consultation available. We respond within 24 hours.
Enter Japan Before Approval
Deliver Your Drug to Patients in Japan — Fast & Compliantly
We support global pharmaceutical and biotech companies in providing access to unapproved medicines in Japan through Early Access Programs, Named Patient Supply, and regulatory-compliant importation.

For Global Pharmaceutical & Biotech Companies
We partner with:
- Pharmaceutical companies
- Biotechnology companies
- Rare disease drug developers
- Specialty pharma companies
- Clinical development companies
Struggling to Supply Drugs in Japan Before Approval?
Japan offers significant opportunities — but entering the market before approval can be complex.
Many global companies face challenges such as:
- Navigating Japan’s strict regulatory framework (PMD Act)
- Lack of a reliable local partner
- Difficulty supplying unapproved medicines
- Limited access to hospitals and physicians
- Complex importation and logistics requirements

We Make Early Access in Japan Simple
Bio Pharmacy provides a complete solution for delivering unapproved medicines to patients in Japan.
We support:
- Early Access Program (EAP)
- Named Patient Program (NPP)
- Compassionate Use / Expanded Access
- Managed Access Program (MAP)
From regulatory compliance to delivery — we handle everything.

Your Trusted Partner in Japan
- Extensive experience in pharmaceutical importation and supply
- Deep expertise in Japan’s regulatory environment (PMD Act)
- Established network with hospitals and medical institutions
- GDP-compliant logistics and temperature-controlled distribution
- Fast, flexible, and reliable execution
We manage the entire process in Japan — so you don’t need a local entity.

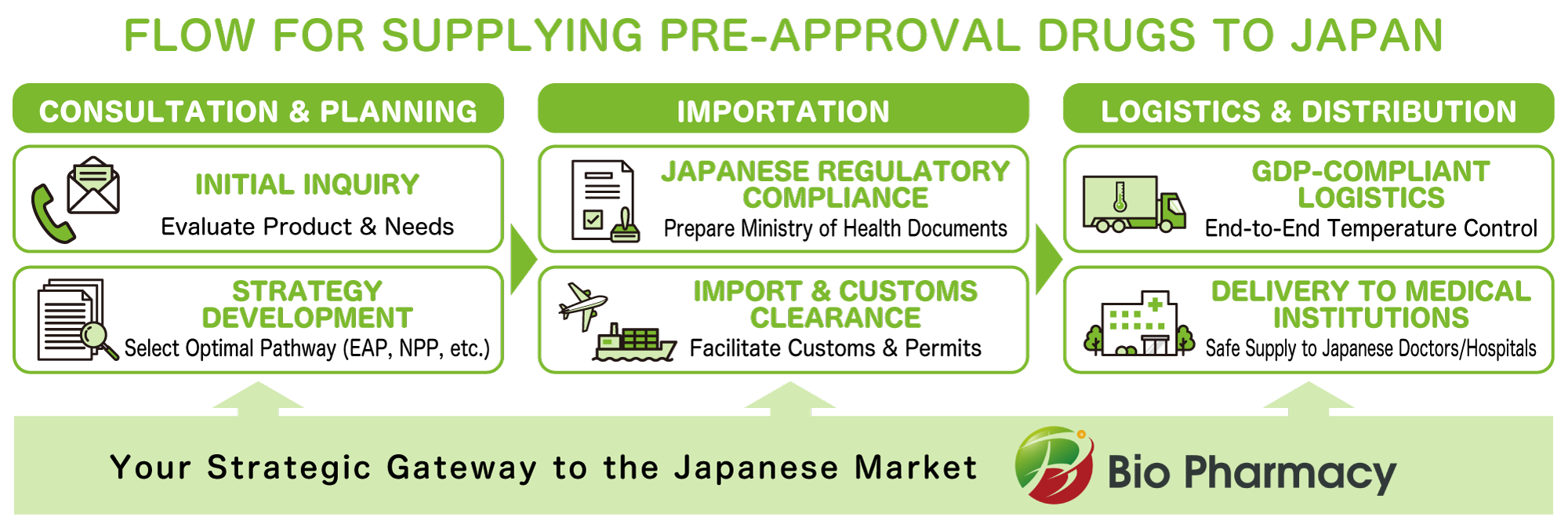
End-to-End Support for Japan Market Access
- Importation support (documentation & regulatory procedures)
- Named patient supply
- Coordination with medical institutions
- GDP-compliant pharmaceutical distribution
- End-to-end logistics management

Why Japan?
Japan is one of the largest pharmaceutical markets in the world, with strong demand for innovative therapies.
Significant unmet medical needs remain in:
- Rare diseases
- Oncology
- Neurology
- Biologics
- Gene therapy
Many treatments approved in the US and Europe are not yet available in Japan, creating a critical opportunity for early access.
Simplifying Access to Japan
- No local entity required
- Full regulatory support provided
- Compliant and secure supply chain
- Support for urgent patient needs
We help bring life-changing treatments to patients in Japan — safely and efficiently.

Let’s Bring Your Innovation to Japan
Partner with Bio Pharmacy to deliver new treatment options to patients in Japan — before approval.
Bio Pharmacy Inc.
2F Otemachi Bldg., 1-6-1 Otemachi, Chiyoda-ku, Tokyo 100-0004, Japan
Tel: +81-3-4500-6971 / Fax: +81-3-4500-6970
